June 20, 2023 Tags: filler injections, under eye bags remedy, fillers for under eye bags
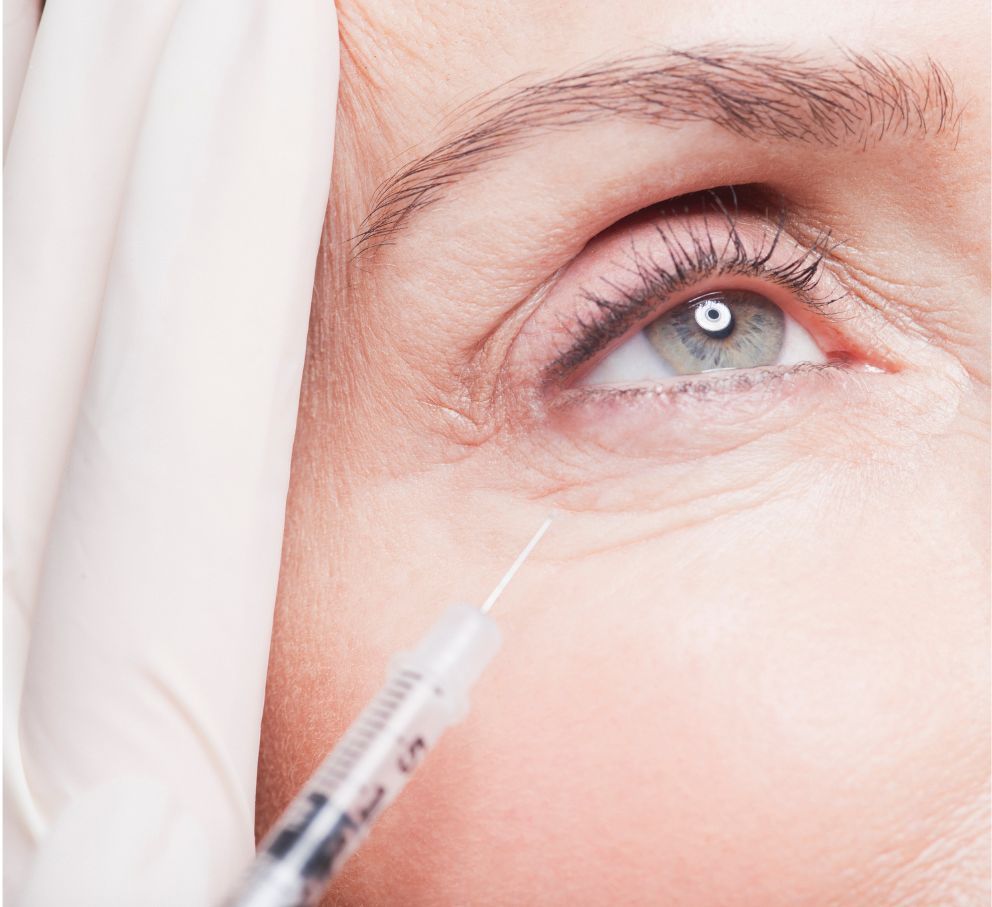
In a breakthrough development for the field of cosmetic dermatology, Alderma has announced that their innovative Restylane Eyelight filler has obtained FDA approval. This new hyaluronic acid filler offers a revolutionary solution for individuals seeking to address under-eye hollows and dark circles.
What sets Restylane Eyelight apart is its utilization of NASHA Technology, making it the first and only dermal filler in the United States to incorporate this cutting-edge advancement. By closely mimicking the body’s natural hyaluronic acid, Restylane Eyelight creates a gel texture that offers optimal support with minimal crosslinking. This unique formulation ensures a more natural-looking result.
During the clinical study process, a notable 87 percent of participants observed a significant decrease in under-eye hollowness within a three-month period. Impressively, the majority of patients, amounting to 92 percent, expressed their satisfaction with the results at the three-month mark. Equally remarkable, an impressive 84 percent reported their continued contentment with the treatment after one year, and an overwhelming 93 percent of individuals expressed a keen interest in undergoing the treatment again after a twelve-month interval.
To learn more about your options for injectable fillers, schedule a consultation with Dr. J.J. Wendel Plastic Surgery today!